Three air molecules go into a hole,
Well, I say three; could have been four or five.
Could have been nine or ten, doesn’t matter.
Could have been fifteen, twenty – fifty.
Round it up. Hundred. Let’s go mad, eh – two-fifty.
…
Alright, make it a lot simpler, all the air molecules on the planet go into a hole, right?
Hang on – they wouldn’t fit would they? What do you think? What proportion of the Earth’s atmosphere could we fit in a hole through the middle of the Earth. The atmosphere, if it was compressed to sea level pressure occupies about 4 billion cubic km, and a hole through the Earth of 1 square metre cross section would occupy about 0.013 cubic km, so even allowing for a fair bit of extra compression, we aren’t going to get much of the atmosphere into the hole are we? Let’s do the numbers.
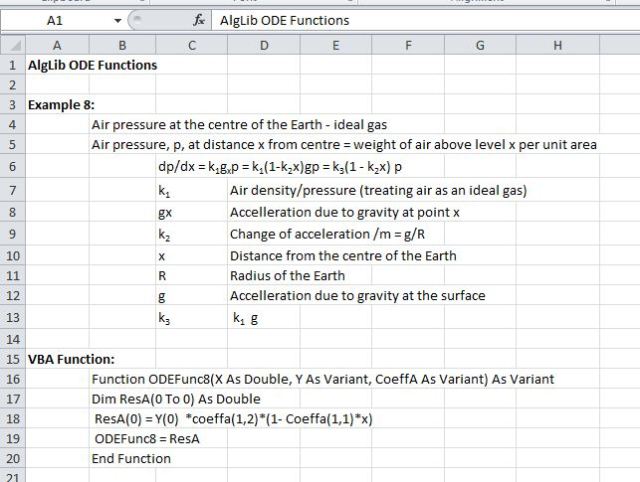
Using the AlgLib ODE solver we can easily write a function to calculate the pressure at any distance from the Earth’s centre, starting at normal pressure (say 100 kPa) at the surface. The ODE and the function code are shown in the screen-shot below.
To keep things simple I have made the following assumptions:
- Air acts as an ideal gas all the way down the hole
- Temperature changes are ignored
- The acceleration due to gravity varies linearly from the surface to zero at the centre
1 and 2 are obviously fairly gross approximations, but we only want an order of magnitude result, so let’s see how they work out.
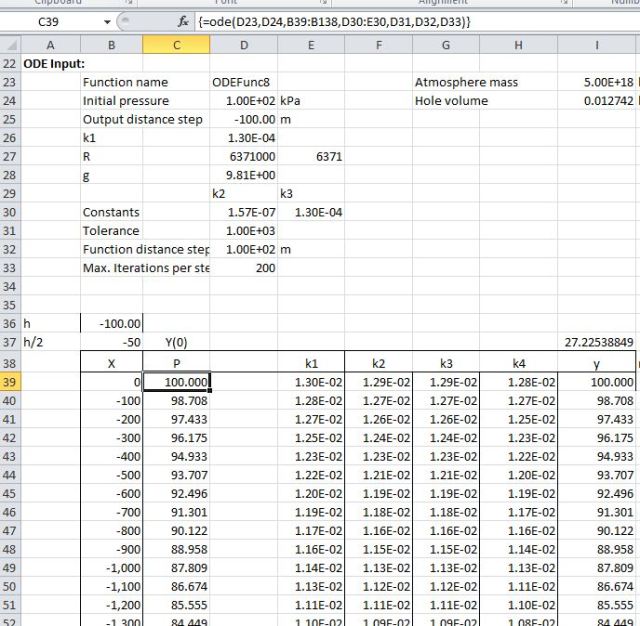
To check out the ODE solver I have started off going from the surface up to an elevation of 10,000 m:
This gives a result of 27.2 kPa, which compares pretty well with the actual pressure (extracted from the Internet) of 26 kPa. Also the ODE function was in close agreement with a simple Runge-Kutta solution carried out on the spreadsheet (see screenshot).
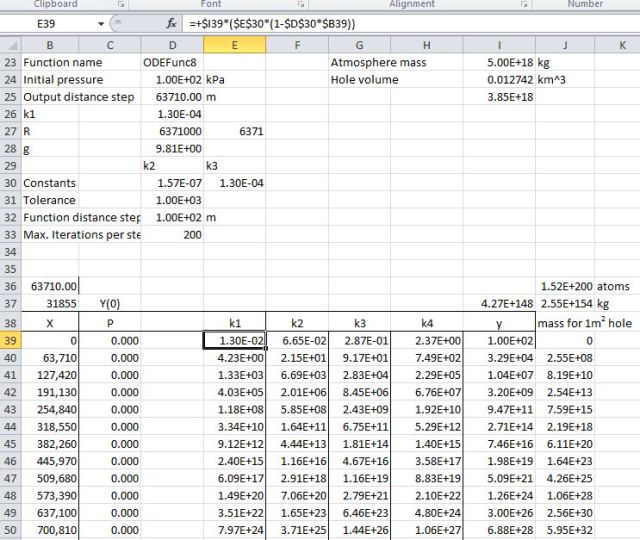
The next step was to go downwards in 100 steps of approximately 64 km each. Here we run into a problem. The AlgLib function fails to converge over this distance. The problem is that it has an iterative error minimisation routine, but very big numbers cause it to crash. To get around this I have used the simple Runge-Kutta method on the spreadsheet, without any error minimisation. Using this we find a pressure at the centre of about 4.3E148 kPa (43 followed by 147 zeros!):
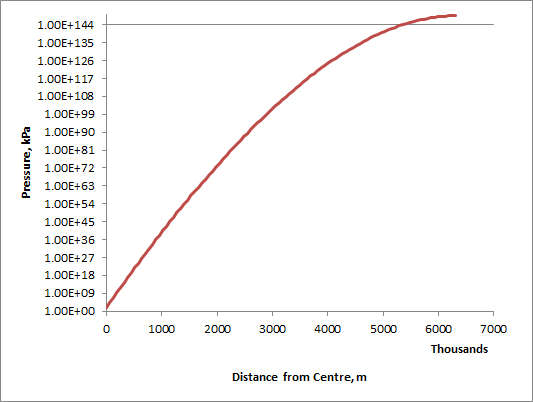
Here is a graph of the pressure against depth on a log vertical scale:
The total mass of air in the hole would be 2.6E164 kg, which is composed of about 1.5E200 atoms.
It is difficult to comprehend just how big these numbers are, but some idea is given by considering that the total number of atoms in the observable universe is estimated to be of the order of 1E80. We would need 1.5E120 Universes to fill the hole. To look at it another way, if every atom in the observable universe was a universe of equal size itself, and if every atom in every one of those universes contained a mass of 200 billion tonnes, then that would provide enough perfect gas to fill the hole so that the pressure at the surface was just 100 kPa.
It is fortunate that no real gas behaves remotely like a perfect gas at high pressures!
In the next post in this series I will look at how much air we would need using real gas behaviour. I just need to find a source of information for compressibility of air at very high pressures.

For real air, the major components will be a supercritical fluid after a couple of hundred km, and beyond that will form various crystalline states.
LikeLike
Pingback: 3 year report | Newton Excel Bach, not (just) an Excel Blog
Pingback: Daily Download 28: Science (and using ODE solvers) | Newton Excel Bach, not (just) an Excel Blog
Pingback: Falling Faster | Newton Excel Bach, not (just) an Excel Blog